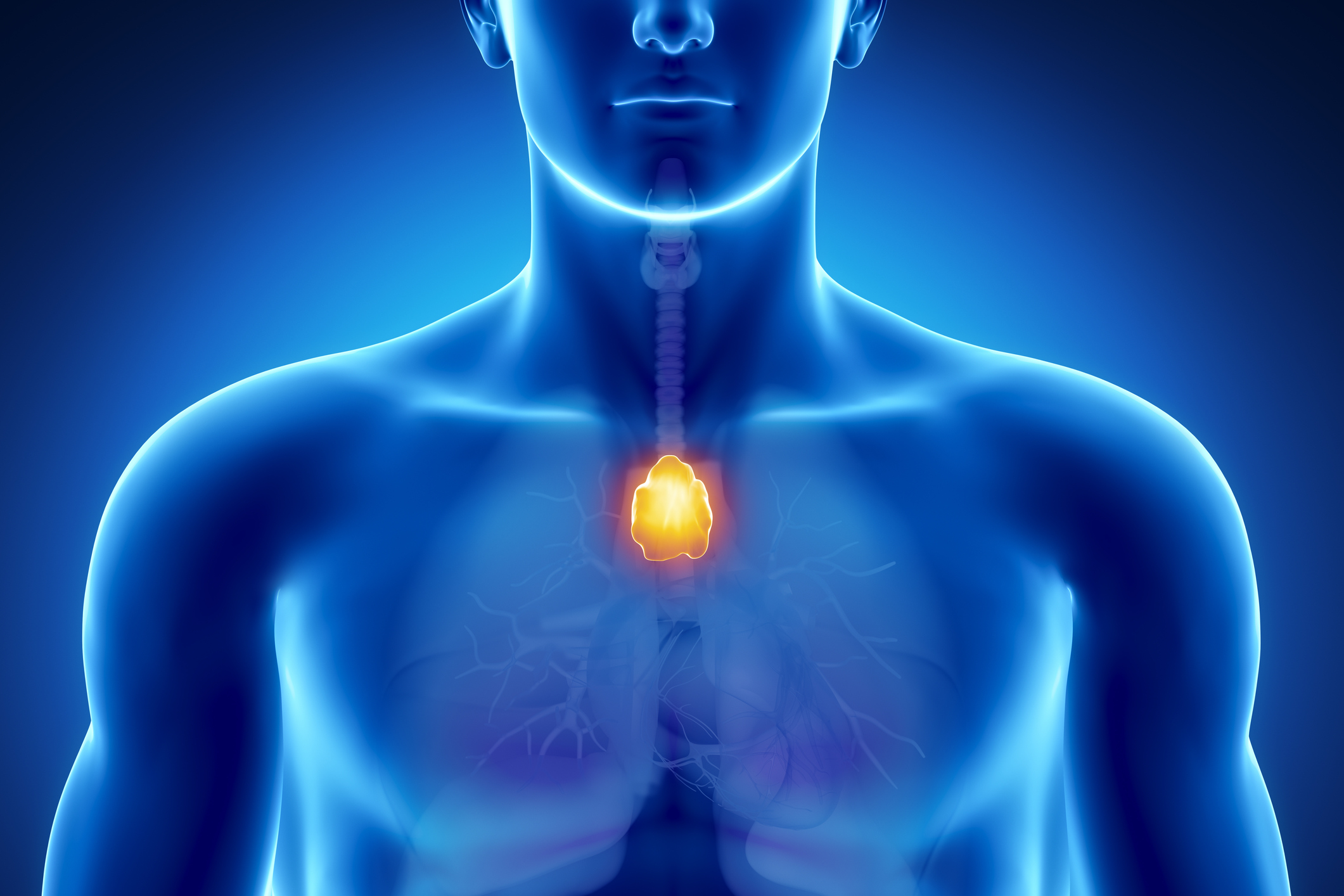
- Myasthenia gravis is an autoimmune disorder causing muscle weakness due to impaired nerve-muscle communication.
- Age and gender are key risk factors for myasthenia gravis, as it often affects younger women (<40) and older men (>60) more.
- Other autoimmune diseases can increase the risk of developing or worsening MG.
- Infections, certain medications, stress, and surgery are common triggers that can exacerbate myasthenia gravis symptoms.
Speak to a Specialist
About Copay AssistanceMyasthenia gravis (MG) is a chronic autoimmune disorder characterized by weakness in the muscles responsible for movement. It is a relatively rare condition, with a prevalence of approximately 150 to 200 cases per million.
What are the risk factors for myasthenia gravis? Common myasthenia gravis risk factors include older age for men, younger age for women, and certain genetic markers.
In this guide, we will dive deeper into these factors. We will explain who is most at risk for myasthenia gravis and discuss the causes of this disease that you should be aware of.
What is Myasthenia Gravis?
Myasthenia gravis (MG) is a chronic autoimmune disorder that causes muscle weakness. This occurs when the communication between nerves and muscles is impaired.
Myasthenia gravis has several symptoms, some of which include:
- Chronic fatigue
- Difficulty walking
- Drooping eyelids (ptosis)
- Trouble holding up the head
- Limited facial expressions
- Double or blurred vision (diplopia)
- Weakness in the neck, arms, or legs
- Problems with swallowing or chewing
- Difficulty speaking (soft or nasal speech)
Although there’s usually no cure, myasthenia gravis has several treatment options that can help improve symptoms.
What Causes Myasthenia Gravis?
The exact causes of myasthenia gravis are unknown. However, health experts believe specific genetic changes may make you more susceptible to developing this condition.
In myasthenia gravis, your immune system produces proteins (antibodies) that disrupt the communication between your nerves and muscles. Why this happens is not well understood.
Several studies have linked MG with thymoma (a rare tumor that forms on the thymus). That said, researchers do not yet know who is most at risk for myasthenia gravis and whether thymoma causes the disease.
What Are the Major Risk Factors for Myasthenia Gravis?
The major risk factors of myasthenia gravis include:
Age
Anyone can get MG, regardless of their age. However, women between 20 and 39 and men between 50 and 70 are more likely to develop this condition [1].
Gender
Like other autoimmune disorders, generalized myasthenia gravis affects more women than men. The female-to-male ratio of MG patients is about 2:1 [2].
Some women may experience their first symptoms during pregnancy. Many women report a worsening of symptoms before their menstrual cycle.
Thyroid

Having a thyroid condition is another risk factor for myasthenia gravis that may make you more likely to develop the disease. About 5% to 10% of people with MG have thyroid disease [3].
Thyroid conditions affect the thyroid gland in the neck, which produces hormones regulating metabolism. Hashimoto’s thyroiditis, which causes hypothyroidism (an underactive thyroid), and Graves’ disease, which results in hyperthyroidism (an overactive thyroid), are the most common conditions associated with MG.
Get Financial Assistance
Other Diseases
In rare cases, other autoimmune disorders may raise your risk of myasthenia gravis. Someone who is diagnosed with the following conditions may be at most risk for developing myasthenia gravis:
- Rheumatoid arthritis (RA): Rheumatoid arthritis is a chronic inflammatory disorder that affects the joints, causing pain, swelling, and stiffness. However, it can also affect other parts, including the skin, eyes, lungs, heart, and blood vessels.
- Systemic lupus erythematosus (SLE): Often called lupus, SLE is a chronic autoimmune disease that can affect various parts of the body, including the joints, skin, kidneys, blood cells, brain, heart, and lungs. It can cause widespread inflammation and tissue damage.
Myasthenia Gravis Risk Factors: Decoding the Role of Genetics
Genetic changes are thought to increase the risk of MG. However, these genes haven’t been identified yet.
Moreover, MG is not typically inherited and can occur in those with no family history. A family history of MG or other autoimmune conditions is present in only about 4% of the cases [4].
Rarely, children born to affected mothers get MG. This is known as neonatal myasthenia gravis. Neonatal MG is temporary. With appropriate treatment, complete recovery occurs within 60 days after birth.
Congenital myasthenic syndrome is a rare group of genetic conditions similar to myasthenia gravis but with different risk factors. It’s present in children at birth, with most symptoms beginning to show in childhood. However, in some cases, symptoms may not show until adulthood. It’s not an autoimmune condition, and its treatment differs from MG.
What Are the Other Potential Risk Factors for Myasthenia Gravis?
A 2023 study found that women over 50 who were diagnosed with type 2 diabetes may be at most risk for developing myasthenia gravis [5].
This research analyzed data from 118 hospitalized MG patients and compared them to several control groups, including the general population and patients with other autoimmune diseases.
The findings indicated that women over age 50 with type 2 diabetes had a significantly higher risk of developing MG. Interestingly, patients with diabetic MG tended to experience onset at an older age compared to those without diabetes.
The study suggests that diabetes may be one of the risk factors for myasthenia gravis, especially in older women. However, these are preliminary results. Further research is needed to confirm the link and understand the underlying mechanisms.
What Factors Can Make Myasthenia Gravis Worse?
If you have MG, certain factors can exacerbate your symptoms or trigger a worsening of your condition. Being aware of these triggers can help you manage your health better and avoid unnecessary complications. Here is an overview of these factors.
Infection
Infections can often worsen the symptoms of myasthenia gravis. When the body fights off infections, it naturally ramps up the immune response. This heightened response can intensify the autoimmune attack on the neuromuscular junction.
Common infections such as respiratory illnesses, urinary tract infections, or even minor colds can lead to increased weakness, fatigue, and in some cases, precipitate a myasthenic crisis.
Prompt treatment of infections and maintaining good hygiene are vital in minimizing this risk. If you notice your symptoms worsening during an infection, consult your healthcare provider immediately.
Get Copay Assistance Now
Immunization
While vaccines are generally safe and essential for preventing serious diseases, certain immunizations can act as risk factors and temporarily exacerbate myasthenia gravis symptoms. Live vaccines or certain adjuvanted vaccines may trigger a flare-up.
The reason someone who is vaccinated is most likely at risk for experiencing a worsening of their myasthenia gravis symptoms is due to the immune system’s response to the vaccine, which can temporarily heighten autoimmune activity.
It’s important to discuss your condition with your healthcare provider before receiving any vaccination. They can advise on the safest options, timing, and whether premedication might be necessary.
In some cases, maintaining vaccination schedules is critical to prevent infections that could be risk factors for myasthenia gravis and worsen the patient’s condition.
Surgery
Surgical procedures, especially those involving anesthesia or significant physiological stress, can impact MG symptoms. Surgery can cause physical stress and immune activation, temporarily worsening muscle weakness.
One treatment option for MG is thymectomy, which involves the removal of the thymus gland. However, this type of surgery requires careful planning before the operation and close monitoring afterward.
Anesthesia agents can also interfere with neuromuscular transmission. In these cases, anesthesiologists experienced with MG must be present during the operation.
If you’re scheduled to undergo surgery, it’s vital to inform your surgical team about your myasthenia gravis diagnosis so they can remedy factors that may risk exacerbating your condition. Proper perioperative management can minimize risks and ensure safer surgery.
Certain Drugs
Certain medications for other health conditions can interfere with neuromuscular transmission and worsen MG symptoms.
What are the drugs that can act as risk factors for exacerbating myasthenia gravis symptoms?
- Certain antimalarial drugs (e.g., quinine and quinidine)
- Seizure medications (e.g., phenytoin, carbamazepine, and phenobarbital)
- Some antibiotics (e.g., aminoglycosides such as gentamicin and amikacin)
- Medications for irregular heart rhythms (e.g., beta-blockers such as propranolol and metoprolol)
- Statins for lowering cholesterol (e.g., atorvastatin, rosuvastatin, simvastatin)
It is best to avoid these drugs if you have MG. However, if needed, they should be used with caution and at the lowest dose possible. They can impair nerve-muscle communication, a factor that may increase the risk of myasthenia gravis symptoms worsening. Ask your doctor to recommend alternatives if possible.
Stress
Someone who is emotionally or physically stressed is also most likely at risk for exacerbating their myasthenia gravis symptoms.
Stress hormones can influence immune function and neuromuscular transmission, leading to increased weakness and fatigue. Prolonged or intense stress may also lower your threshold for experiencing a myasthenic crisis.
Developing stress management techniques such as relaxation exercises, mindfulness, and adequate rest can help reduce this risk. Maintaining a stable routine and seeking emotional support when needed are also helpful strategies to control your symptoms.
Myasthenia Gravis Risk Factors for Recurrence: What You Should Know
In some cases, the symptoms may reappear in people with MG who have had a thymectomy (surgery to remove the thymus). The risk is higher in [6]:
- Males
- Older adults
- People with more severe disease
Death risk in MG patients may be higher if they have more severe symptoms, a stroke, diabetes, higher cholesterol levels, and abnormal heartbeats [7].
Speak to a Specialist
About Copay AssistanceMyasthenic Crisis Risk Factors

Myasthenic crisis (MC) is a life-threatening complication of MG. It causes your breathing muscles to become too weak. You may need a breathing machine (ventilator) in severe cases.
MC is more likely to occur if you have [8]:
- Thymoma
- Higher antibody levels
- Severe symptoms at diagnosis
Manage MG With AmeriPharma® Specialty Pharmacy
Understanding the risk factors for myasthenia gravis can help you better manage your condition and avoid complications. If you’re someone who is most at risk for developing myasthenia gravis, you might want to keep a close eye on emerging symptoms.
If you’re already diagnosed with MG and need treatment, AmeriPharma® Specialty Pharmacy can help. Our ACHC-accredited specialty pharmacy provides hard-to-find medications for those dealing with complex conditions like myasthenia gravis.
Contact us today to speak to a patient navigator and start receiving MG treatment with full-service coordination, copay assistance, and 24/7/365 support.


 Symptoms may appear a few weeks after you start taking a drug. Initially, you may have flu-like symptoms, such as:
Symptoms may appear a few weeks after you start taking a drug. Initially, you may have flu-like symptoms, such as:


 According to the National Hemophilia Foundation,
According to the National Hemophilia Foundation,