People with hemophilia experience nearly 30 bleeding events per year. Of these, about 10% involve the mouth, tongue, and gums [1].
Bleeding gums and post-extraction bleeding are the characteristic features of hemophilia. The more severe the condition, the more frequent the bleeding episodes. In children, ulcers and bruises may form on the lips and tongues.
Poor oral hygiene is associated with a higher risk of oral bleeding in people with hemophilia. In this article we will discuss hemophilia and dental treatment in depth.
Top 10 Tips To Prevent Bleeding Gums and Mouth Injuries
The World Federation of Hemophilia recommends the following tips to help reduce the need for treatment and emergency department visits [2]:
- Brush your teeth (for two minutes per session) at least twice daily with fluoride-containing toothpaste.
- Replace your toothbrush at least every 3 months.
- Use mouthwashes containing triclosan or chlorhexidine.
- Choose a brush with medium-texture bristles. Hard bristles can cause mouth injuries, and soft bristles don’t help remove plaques.
- Use interdental cleaning aids — floss, tape, and interdental brushes — to prevent tooth decay and other dental problems.
- You may use fluoride supplements like gels or tablets if the fluoride content of your water supply is less than 1 ppm. Talk with your dentist to learn about ppm and whether you need a fluoride supplement.
- Limit foods and drinks high in sugar and acid to three exposures per day. Examples include carbonated beverages, fruit juices, and cereals. Keep a food diary to track your eating habits.
- You may use artificial sweeteners, such as aspartame, acesulfame, and sorbitol.
- Visit your dentist regularly, preferably every 6 months.
- Quit smoking. You can find help at CDC.gov/quit and Smokefree.gov.
If you or anyone you love has hemophilia, click here to learn how to brush and floss.
Hemophilia and Dental Treatment
Dental treatments can vary depending on the severity of the condition.
For example, if you have mild hemophilia, you will likely receive preventive care in a primary care setting. Preventive measures include:
- Maintaining proper oral hygiene
- Eating a healthy diet
- Sealing pits and fissures
- Fluoride treatments
On the other hand, moderate and severe cases are typically managed in a secondary care setting. You may receive clotting factors before a dental procedure or surgery.
Regardless of the disease severity, your dentist will work closely with a hematologist.
Before any dental procedure, your dentist will assess your medical and dental history. Also, they will ask questions about:
- Other co-occurring medical conditions
- Medications you take
- Your response to past dental treatment
With this information and after consultation with a hematologist, they will design a treatment plan relevant to your needs.
Treatment of Gum Disease

Scaling (above or below the gumline) is the primary treatment for gum disease. You may be prescribed chlorhexidine mouthwash to control oral bacteria growth. Antibiotics may be necessary to treat or prevent infection and reduce swelling of the gums.
Use of Dentures
People with hemophilia can use full or partial dentures (false teeth) as long as they are comfortable.
Use of Braces
With proper preventive measures, people with hemophilia can use fixed or removable braces without any issues.
Tooth Extraction in People with Hemophilia
Tooth extraction is an invasive process. Depending on the disease severity and the number of teeth to be extracted, you may need replacement clotting factors before the procedure.
After your tooth has been extracted, you should avoid rinsing, smoking, or strenuous activities for 24 hours. Also, you shouldn’t take pain medications that increase bleeding risk, such as aspirin.
Fillings, Crowns, and Bridges
These restorative treatments are considered low-risk. Thus, they may be carried out in general dental practice.
Root Canal Treatment
Root canal treatment doesn’t appear to cause additional bleeding problems in people with hemophilia.
Get Financial Assistance
Hemophilia and Dental Treatment: Frequently Asked Questions
How do you stop gum bleeding from hemophilia?
Tranexamic mouthwash and tablets are effective in stopping gum bleeding in hemophilia.
What happens if I need a tooth extraction?
Tooth extraction is an invasive process. You may need replacement clotting factors or other medications before the procedure. You will also need to follow your dentist’s instructions after the procedure.
What should I do if brushing makes my gums bleed?
First, talk to your dentist and follow their instructions, as it could be a sign of gum disease. Continue to brush your teeth (for two minutes per session) at least twice daily with fluoride-containing toothpaste. Use a brush with medium-texture bristles.


![What Is Advate (Antihemophilic Factor [Recombinant])?](https://ameripharmaspecialty.com/wp-content/uploads/2024/05/boy-bandage.jpg)
 Side effects can be mild or severe.
Side effects can be mild or severe. 


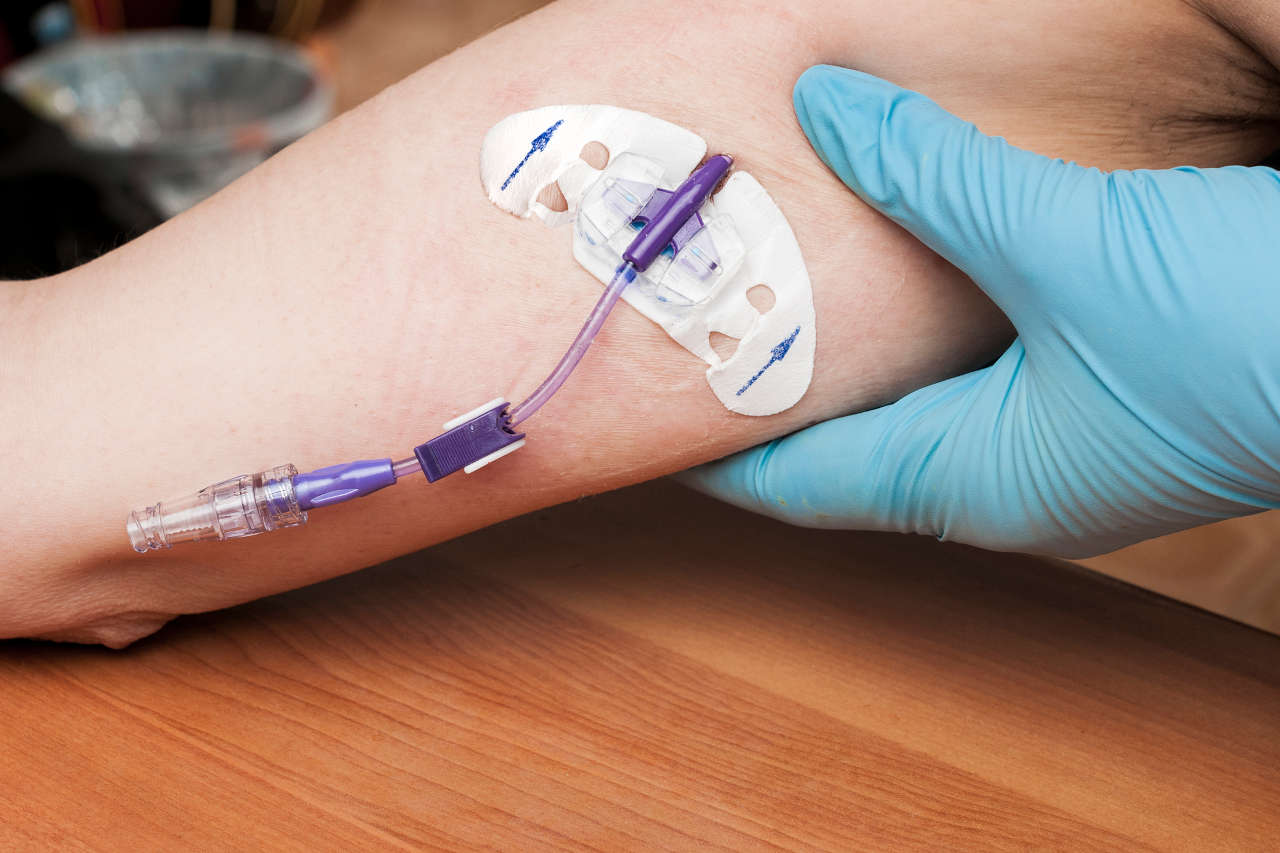
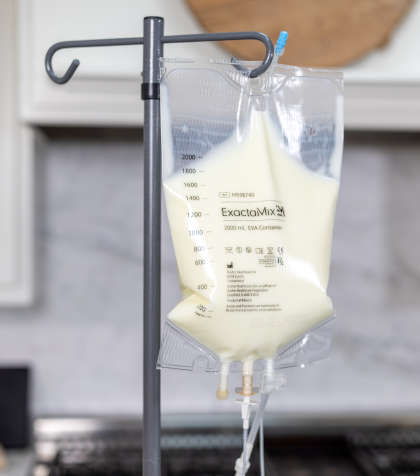

 While subjective experience of well-being is a strong indicator to tell if IVIG is working, several other objective indicators do the same, perhaps in a more reliable way. These include:
While subjective experience of well-being is a strong indicator to tell if IVIG is working, several other objective indicators do the same, perhaps in a more reliable way. These include: