What Is Parenteral Nutrition (PN)?
PN is the medical abbreviation for parenteral nutrition, also known as intravenous nutrition. It is delivered directly to the body through the veins when a person cannot obtain adequate nutrition through their digestive system. When properly managed, this can be safely done at home. There are two subtypes of PN: peripheral parenteral nutrition (PPN) and total parenteral nutrition (TPN).
Your IV Fluids, Always Available
Full inventory, hassle-free accessTotal Parenteral Nutrition (TPN)
Those who cannot obtain nutrition through oral consumption may be prescribed TPN. TPN is typically composed of some or all of the following ingredients: carbohydrates, proteins, lipids, electrolytes, trace elements, and insulin. Trace elements (copper, manganese, zinc, and selenium) and multivitamins may be added for further nutritional supplementation.
Common conditions that TPN is used to manage are:
- Ischemic bowel disease
- Short bowel syndrome
- Cancer
- Inflammatory bowel disease
Generally, patients with conditions that do not allow for a normally functioning digestive tract may benefit from TPN therapy. This includes those who have poor blood flow to their bowels as well.
TPN is usually formulated to meet and replace complete daily nutritional needs. Because TPN solutions can be very concentrated to deliver enough nutrients, a central line, which is an intravenous catheter that accesses one of the largest veins in the body, is typically needed for administration.
Peripheral Parenteral Nutrition (PPN)
PPN is parenteral nutrition administered through a peripheral intravenous line that’s inserted to the neck or one of the limbs. Because it is entered into smaller veins, PPN solutions are much more diluted vs. TPN solutions to prevent extravasation (medication leakage from the vein into the surrounding tissue) and thrombophlebitis (an inflammatory process that causes a blood clot to form and block the vein).
As a result, PPN solutions generally provide significantly fewer calories than TPN solutions. PPN is prescribed for short-term nutritional support, usually for 2 weeks or less.
PPN vs TPN
TPN and PPN both serve the same general purpose; to provide nutrition to the body through alternative means. The main differences between PPN and TPN infusions are associated with calorie content and treatment duration.
PPN is reserved for the short-term when the expected duration of treatment is no greater than 2 weeks, usually during or after an acute event. Solutions are much more dilute and often meant to supplement nutritional needs rather than replace daily requirements.
TPN, on the other hand, is much more comprehensive. Each formula is calorie-dense and may replace the daily needs completely. It is often prescribed over PPN when the condition is expected to be chronic and has no definite recovery time. When PPN treatment extends beyond 2 weeks, it is usually converted to TPN.
Speak to a Specialist
About Copay AssistanceComposition
Total parenteral nutrition usually contains a combination of the following:
- Minerals
- Electrolytes
- Trace elements
- Lipid emulsions
- Proteins/amino acids
- Carbohydrates/dextrose
TPN nutrition composition should be personalized to meet each patient’s specific needs.
Lipid emulsions, amino acids, and dextrose are the three main macronutrients that provide the daily calorie needs.
Carbohydrates:
- Dextrose content in a TPN solution may be anywhere from 10% to 25% (wt/vol). It is often the largest calorie contribution source.
- For most patients, the body’s maximum rate of glucose utilization is between 4 and 5 mg/kg/min. In both TPN and PPN treatments, the dextrose infusion rate is best maintained less than or within this range, depending on the caloric need.
- Carbohydrate over-administration can cause dangerous blood sugar and lipid imbalances.
- Long-term rapid administration of carbohydrates may lead to the development of a fatty liver.
Proteins:
- A healthy adult usually requires between 0.8 and 1 gram of protein per kilogram of body weight per day.
- Patients with acute hepatic encephalopathy require a temporary protein restriction of 0.8 gm/kg/day.
- Patients with critical illnesses may sometimes require up to 2 gm/kg/day.
- Hemodialysis patients generally need 1.2 – 1.3 gm/kg/day.
Lipid Emulsions
- Lipids provide healthy calories and essential fatty acids. Without them, deficiencies can occur after 3 weeks of fat-free TPN nutrition.
- Cholesterol and lipids usually account for 20% to 30% of total calories consumed.
- Lipids can be infused by themselves or be added to the overall solution. When combined, the resulting product is known as a “total nutrient admixture (TNA)” or “3-in-1” TPN (versus “2-in-1” TPN, which contains only amino acids and dextrose). Lipids are what sometimes give these solutions the “milky” color.
Micronutrients: Electrolytes, Vitamins, and Trace Elements
- Essential vitamins are provided as an additive to TPN. They are available as a multivitamin solution created for intravenous administration.
- Trace elements (zinc, copper, manganese, and selenium) are added based on daily needs.
- In most cases, each daily TPN bag will contain the following amounts of electrolytes per liter of solution:
- Calcium (Ca): 5 – 15 mEq/day
- Sodium (Na): 1 – 2 mEq/kg/day
- Potassium (K): 1 – 2 mEq/kg/day
- Magnesium (Mg): 8 – 20 mEq/day
- Phosphorus (Phos): 15 – 30 mM/day
*Exact amounts will vary from patient to patient based on laboratory response.
Ask About TPN At-Home Infusion
TPN Administration
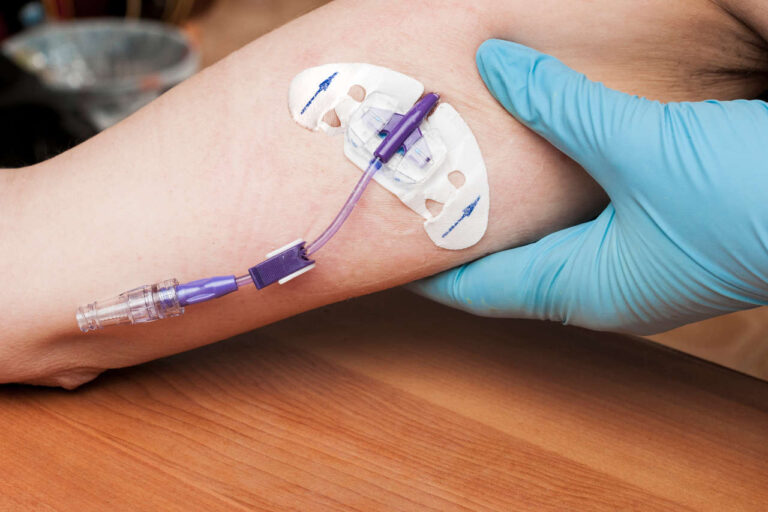
Due to their varied nutrient concentrations, PPN vs. TPN solutions employ different administration methods. Total parenteral nutrition is usually administered via a central venous catheter (CVC). Common types of CVC are peripherally inserted central catheter (PICC line), tunneled CVC, and implanted port. These are used to access the central circulatory system, where more concentrated solutes can be infused.
PICC Lines
PICC lines can be placed in the median cubital veins (brachial, cephalic, or basilic), depending on size and desired location. The catheter is inserted peripherally. It is guided by radiographic imaging until the tip is placed near the superior vena cava of the heart.
PICC lines can be used for TPN infusion for up to 6 months as long as the catheter and the site of insertion remain stable and infection-free. After 6 months, these catheters will need to be replaced with a new one. Upon completion of treatment, PICC lines can safely be removed by a trained nurse in almost any setting.
Tunneled CVCs
Tunneled CVCs are surgically placed to enter the central circulatory system via the internal jugular veins, subclavian, or femoral vein. These function the same way as PICC lines, except they are sutured into the skin. For removal, the patient must be referred back to interventional radiology.
Implanted Ports
An implanted port, commonly known as medi-port or port-a-cath, is the most permanent type of central venous catheter. It is also surgically placed, but the entry point goes under the skin. Each access will require a particular type of needle called a Huber needle, which comes in various sizes depending on preference and how the port was placed.
TPN has long been delivered via implantable ports, especially to patients who require chronic IV nutrition. Ports may stay placed indefinitely as long as the catheter remains stable and infection-free. Removing ports also requires a referral to interventional radiology.
PPN Administration
TPN may not be administered via peripheral intravenous catheter due to high osmolarity. For peripheral administration (PPN), PN solutions must be formulated below 900 mOsm. This provides much less flexibility on how many calories to add to each bag. Therefore, PPN often will not provide enough to meet daily nutritional requirements.
PPN vs. TPN solutions are formulated for different medical needs. PPN is usually reserved for nutritional support rather than nutritional replacement. It is given short-term when the expected need does not exceed 2 weeks. Also, because peripheral catheters access the circulatory system through smaller veins, these devices cannot remain stable for very long after access.
AmeriPharma® Specialty Care
Leader In TPN AssistanceIndications
TPN vs. PPN treatments have different use cases. TPN therapy is considered when a patient’s gastrointestinal function is inadequate and enteral nutrition is contraindicated. Enteral diets are preferable to parenteral diets because they are less expensive and have fewer potential complications. However, it is only possible if the patient has a functional gastrointestinal system.
A patient’s condition and symptoms will determine whether they need PPN vs. TPN. The following indications necessitate TPN feeding:
- Cancer of the digestive tract: Tumors can obstruct the bowels and compromise the consumption of sufficient calories. Chemotherapy and other cancer treatments may also cause your body difficulty absorbing nutrients.
- Food intolerance combined with pseudo-obstruction of the bowel.
- GI fistulas with high flow: TPN nutrition can be used to rest the bowel and replace fluids lost from drains.
- Pediatrics: Children and infants may have immature digestive tracts or congenital gastrointestinal malformation.
- A leak from a postoperative bowel anastomosis.
- Severe vomiting or diarrhea that prevents adequate nutritional intake.
- Small bowel obstruction.
- Hypercatabolic states such as sepsis, polytrauma, significant fractures, and burns.
- Exacerbations of inflammatory bowel disease.
- Critically ill patients.
- Other conditions with expected NPO (nothing by mouth) time longer than 7 days.
Other Reasons for TPN
The following conditions may require the use of TPN depending on their severity:
Crohn’s Disease
Crohn’s disease is an inflammatory bowel disease that may manifest as abdominal pain, bowel narrowing, and other symptoms that impair food intake, digestion, and absorption.
Short bowel syndrome
A person may be born with this condition or develop it due to any surgery removing a significant portion of the small intestine. A patient with short bowel syndrome does not have enough bowels to absorb adequate nutrients from oral consumption.
Ischemic bowel disease
In ischemic bowel disease, bowel dysmotility and malabsorption result from decreased blood flow to the intestines due to cardiovascular causes.
Other abnormal bowel function
Idiopathic abnormal bowel function is caused by disruptions in bowel motility due to external factors. Some causes include surgical adhesions, radiation enteritis, and small fiber neuropathy.
Speak to a Specialist
About Copay AssistanceTPN and PPN in Pregnancy
Nausea and vomiting are widespread side effects of pregnancy, and they can significantly impair activities of daily living, including nutrition. Approximately 85% of pregnant women experience nausea and vomiting during the first 3 months of pregnancy. 20% may continue to have them for the remainder of the pregnancy.
Approximately 2% of pregnant women suffer from hyperemesis gravidarum, a severe form of nausea and vomiting that can last for several days and even weeks. This high-risk condition may put both the mother and the fetus in danger. According to the American College of Obstetricians and Gynecologists (ACOG), parenteral nutrition may be considered in pregnant patients who have lost 10% or more of their pre-gestation body weight.
Whether a patient needs PPN vs. TPN treatment will depend on the severity of their condition. Treatment may be continued until birth if the mother continues to have difficulty tolerating oral intake despite using antiemetics. Complications from hyperemesis gravidarum, such as ketonuria, severe dehydration, and catabolism, are emergencies. They may necessitate hospitalization.
Contraindications
According to Maudar (1995), total parenteral nutrition is generally contraindicated in the following circumstances:
- Infants that have a small bowel with less than 8 cm in length.
- Patients who are irreversibly decerebrate.
- Patients who are suffering from severe cardiovascular instability or metabolic instabilities. Before initiating parenteral nutrition, it is necessary to correct any instabilities.
- The patient’s digestive tract is functional and usable.
- The patient’s nutritional status is favorable with only short-term expected benefits from TPN.
- Patient death is unavoidable regardless of nutritional status. Total parenteral nutrition should not be used to prolong life in such situations.
Ask About TPN At-Home Infusion
TPN Monitoring
Delivering complete nutrition makes TPN risker vs. PPN treatments. As a result, the following parameters are routinely monitored during TPN therapy:
- Liver function
- Serum protein
- Urine sugar levels (usually in the hospital)
- Input and output charts (usually in the hospital)
- Blood urea nitrogen (BUN) and serum creatinine
- Serum electrolytes: sodium, potassium, chloride, bicarbonate, calcium, magnesium
At PN initiation, patients should be monitored closely for signs or symptoms of refeeding syndrome, which manifests as severe electrolyte instabilities, mainly with potassium, magnesium, and phosphate.
The risk is greatest with severely malnourished and cachectic individuals (those who have wasting syndrome). Patients experiencing refeeding syndrome may also present with respiratory distress, acute kidney injury, and even rhabdomyolysis (which is the breakdown of muscle tissue that releases a damaging protein into the blood). A slow initial TPN infusion and titration, especially with dextrose and lipids, will help prevent refeeding syndrome.
Complications
TPN therapy does not come without risks. Some possible major adverse events to consider are:
- Edema
- Infection
- Bleeding
- Air embolism
- Vascular injury
- Pneumothorax (collapsed lung)
- Liver dysfunction
- Venous thrombosis
- Chemically-induced metabolic abnormalities
Catheter-Related Infections
Unfortunately, TPN and PPN treatments also encourage the growth of other unwanted organisms, such as bacteria and fungi. Patients on chronic TPN therapy are at higher risk of the following complications associated with intravenous catheters:
- Skin infection at the site of the catheter insertion
- Bloodstream infections, also known as bacteremia or sepsis
With proper aseptic and sterilization techniques, these risks can be significantly minimized. Consult your nurse on how to best care for intravenous catheters.
AmeriPharma® Specialty Care
Total Parenteral NutritionMetabolic Abnormalities:
Because complete nutrition and electrolytes are administered directly into the veins, patients on TPN are more susceptible to metabolic fluctuations vs. PPN therapy, especially when solutions are administered too quickly. Common metabolic complications with TPN therapy include:
- Refeeding syndrome: The biggest concern with this is at the beginning of TPN therapy, especially with patients who have severe malnutrition. The risk is minimized with the slow titration of calories to the desired goal.
- Hyperglycemia: This is most concerning in patients with diabetes.
- Hypoglycemia: This risk is increased with abrupt discontinuation of infusions. To minimize, TPN and PPN infusions should be tapered up at the start and tapered down at the end.
- Electrolyte abnormalities: This is regularly evaluated via labs. It is important to have blood draws done at least once weekly while on TPN therapy to prevent imbalances.
- Cholestasis: This is a consequence of prolonged bowel rest. If the patient has somewhat of a functional GI tract, they should be encouraged to have some oral intake, no matter how small. If NPO (nothing by mouth), the patient should be regularly evaluated for this possibility.
Metabolic Bone Disease
Some patients who have been on TPN nutrition for more than 3 months develop metabolic bone disease, also known as bone demineralization (osteoporosis or osteomalacia). The cause of this is not well defined. Advanced forms of this disease can cause severe back, lower extremity, periarticular pain, swelling, and inflammation.
TPN-Induced Liver Toxicity
Excess caloric intake with glucose and lipids can lead to hepatic steatosis. Its common cause for most patients is a glucose infusion rate of more than 5 mg/kg/min. High blood glucose and insulin levels trigger hepatic lipogenesis. To avoid this, the dextrose dose should be reduced to less than 5 g/kg per day with an infusion rate of less than 5 mg/kg/min.
Liver dysfunction may occur without regard to caloric intake. The risk becomes more significant the longer a patient is on TPN vs. PPN therapy. Liver function tests, including transaminases, bilirubin, and alkaline phosphatase, are monitored to detect signs of liver dysfunction.
Volume Overload
Patients who need a high number of calories often also require a large volume of fluids. In these cases, they are prone to volume overload, which is indicated by a weight gain of more than 1 kg/day. Symptoms include edema, shortness of breath, and frequent urination. This risk is less likely in PPN vs. TPN treatments.
Speak to a Specialist
About Copay AssistanceTPN Healthcare Team
A well-coordinated health care team using an interprofessional approach is required to manage TPN therapy.
Teams usually include:
- Dietician (RD)
- Infusion nurse (RN)
- Pharmacist (PharmD/RPh)
- Prescriber (MD, DO, NP, or PA)
The prescriber decides on the appropriate course of treatment and the type of nutrition. Collaborating with the patient’s primary health care team, the clinician ensures that care is coordinated appropriately.
The pharmacist provides sterile parenteral nutrition. Consult a pharmacist about the compound’s stability and any possible drug/nutrient interactions. Under the prescriber’s directions, pharmacists may make reasonable adjustments to the TPN formula based on clinical assessments and laboratory results.
The dietician assesses the patient’s nutritional status, calculates the daily requirements, and designs an appropriate feeding regimen.
The TPN nursing specialist oversees catheter and TPN tube care. They educate and train the patient and the caregiver on adequately managing the TPN tubes in their homes. The extended team of professionals may also include wound management nurses, occupational therapists, and social workers.
What is the Prognosis for Those on PN?
According to ASPEN’s Parenteral Nutrition Fact Sheet, children and adults can thrive on parenteral nutrition when no complications arise. Generally, the goal of TPN and PPN infusions is to continue until the patient can maintain weight and energy by oral intake alone. Doctors may do a trial of holding PN therapy for several days before discontinuing it. Depending on the indication, the total duration may be anywhere from just a few weeks to several years.
Can You Eat While Receiving TPN?
Depending on the condition and the reason for receiving TPN, you can consume food and beverages while on the therapy. If it is a possibility, oral intake is often encouraged to help expedite discontinuation. However, this should still be done cautiously under the direction of the prescriber and/or dietician because early oral intake may sometimes worsen the condition.
Ask About TPN At-Home Infusion
TPN Price
Without insurance coverage of any kind, the cash price for 500 ml of TPN containing a basic mixture of amino acids, dextrose, and electrolytes is approximately $176 per day. The price may vary depending on the pharmacy. Additional additives may alter this estimate as well.
| Quantity | Per unit | Price |
|---|---|---|
| 500 (25 x 20 milliliters) | $0.35 | $176.33 |
REFERENCES:
- Braunschweig C, Liang H, Sheean P. Indications for administration of parenteral nutrition in adults. Nutr Clin Pract. 2004 Jun;19(3):255-62. doi: 10.1177/0115426504019003255. PMID: 16215113.
- Chowdary KV, Reddy PN. Parenteral nutrition: Revisited. Indian J Anaesth. 2010 Mar;54(2):95-103. doi: 10.4103/0019-5049.63637. PMID: 20661345; PMCID: PMC2900762.
- Weimann A, Ebener Ch, Holland-Cunz S, Jauch KW, Hausser L, Kemen M, Kraehenbuehl L, Kuse ER, Laengle F; Working group for developing the guidelines for parenteral nutrition of The German Association for Nutritional Medicine. Surgery and transplantation – Guidelines on Parenteral Nutrition, Chapter 18. Ger Med Sci. 2009 Nov 18;7:Doc10. doi: 10.3205/000069. PMID: 20049072; PMCID: PMC2795372.
- Messing B. Nutrition parentérale: indications et techniques [Parenteral nutrition: indications and techniques]. Ann Med Interne (Paris). 2000 Dec;151(8):652-8. French. PMID: 11173709.
- Maudar KK. TOTAL PARENTERAL NUTRITION. Med J Armed Forces India. 1995 Apr;51(2):122-126. doi: 10.1016/S0377-1237(17)30942-5. Epub 2017 Jun 26. PMID: 28769264; PMCID: PMC5529889.
- Gotthardt DN, Gauss A, Zech U, Mehrabi A, Weiss KH, Sauer P, Stremmel W, Büchler MW, Schemmer P. Indications for intestinal transplantation: recognizing the scope and limits of total parenteral nutrition. Clin Transplant. 2013 Jul-Aug;27 Suppl 25:49-55. doi: 10.1111/ctr.12161. PMID: 23909502.
- Slattery E, Rumore MM, Douglas JS, Seres DS. 3-in-1 vs 2-in-1 parenteral nutrition in adults: a review. Nutr Clin Pract. 2014 Oct;29(5):631-5. doi: 10.1177/0884533614533611. PMID: 25606645.
- Mohandas KM, Shastri YM, Shirodkar M. Total parenteral nutrition. Natl Med J India. 2003 Jan-Feb;16(1):29-33. PMID: 12715955.
- Wischmeyer PE. The glutamine debate in surgery and critical care. Curr Opin Crit Care. 2019 Aug;25(4):322-328. doi: 10.1097/MCC.0000000000000633. PMID: 31247630.
- Tsujimoto T, Shimizu K, Hata N, Takagi T, Uejima E, Ogura H, Wasa M, Shimazu T. Both high and low plasma glutamine levels predict mortality in critically ill patients. Surg Today. 2017 Nov;47(11):1331-1338. doi: 10.1007/s00595-017-1511-0. Epub 2017 Apr 3. PMID: 28374265.
- Heyland D, Muscedere J, Wischmeyer PE, Cook D, Jones G, Albert M, Elke G, Berger MM, Day AG; Canadian Critical Care Trials Group. A randomized trial of glutamine and antioxidants in critically ill patients. N Engl J Med. 2013 Apr 18;368(16):1489-97. doi: 10.1056/NEJMoa1212722. Erratum in: N Engl J Med. 2013 May 9;368(19):1853. Dosage error in article text. PMID: 23594003.
- Kovacevich DS, Corrigan M, Ross VM, McKeever L, Hall AM, Braunschweig C. American Society for Parenteral and Enteral Nutrition Guidelines for the Selection and Care of Central Venous Access Devices for Adult Home Parenteral Nutrition Administration. JPEN J Parenter Enteral Nutr. 2019 Jan;43(1):15-31. doi: 10.1002/jpen.1455. Epub 2018 Oct 19. PMID: 30339287.
- Gonzalez R, Cassaro S. Percutaneous Central Catheter. 2020 Sep 7. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 Jan–. PMID: 29083596.
- Leib AD, England BS, Kiel J. Central Line. 2020 Jul 31. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 Jan–. PMID: 30137796.
- Parienti JJ, Mongardon N, Mégarbane B, Mira JP, Kalfon P, Gros A, Marqué S, Thuong M, Pottier V, Ramakers M, Savary B, Seguin A, Valette X, Terzi N, Sauneuf B, Cattoir V, Mermel LA, du Cheyron D; 3SITES Study Group. Intravascular Complications of Central Venous Catheterization by Insertion Site. N Engl J Med. 2015 Sep 24;373(13):1220-9. doi: 10.1056/NEJMoa1500964. PMID: 26398070.
- Mattioli S, Miglioli M, Montagna P, Lerro MF, Pilotti V, Gozzetti G. Wernicke’s encephalopathy during total parenteral nutrition: observation in one case. JPEN J Parenter Enteral Nutr. 1988 Nov-Dec;12(6):626-7. doi: 10.1177/0148607188012006626. PMID: 3148047.
- Ayers P, Adams S, Boullata J, Gervasio J, Holcombe B, Kraft MD, Marshall N, Neal A, Sacks G, Seres DS, Worthington P; American Society for Parenteral and Enteral Nutrition. A.S.P.E.N. parenteral nutrition safety consensus recommendations. JPEN J Parenter Enteral Nutr. 2014 Mar-Apr;38(3):296-333. doi: 10.1177/0148607113511992. Epub 2013 Nov 26. PMID: 24280129.
- Lakananurak N, Gramlich L. The Role of Preoperative Parenteral Nutrition. Nutrients. 2020 May 6;12(5):1320. doi: 10.3390/nu12051320. PMID: 32384662; PMCID: PMC7285090.
- Guglielmi FW, Boggio-Bertinet D, Federico A, Forte GB, Guglielmi A, Loguercio C, Mazzuoli S, Merli M, Palmo A, Panella C, Pironi L, Francavilla A. Total parenteral nutrition-related gastroenterological complications. Dig Liver Dis. 2006 Sep;38(9):623-42. doi: 10.1016/j.dld.2006.04.002. Epub 2006 Jun 12. PMID: 16766237.
- Nightingale J. Nutrition support teams: how they work, are set up and maintained. Frontline Gastroenterol. 2010 Oct;1(3):171-177. doi: 10.1136/fg.2009.000224. Epub 2010 Aug 5. PMID: 28839571; PMCID: PMC5517177.
- Russo-Stieglitz KE, Levine AB, Wagner BA, Armenti VT. Pregnancy outcome in patients requiring parenteral nutrition. J Matern Fetal Med. 1999 Jul-Aug;8(4):164-7. doi: 10.1002/(SICI)1520-6661(199907/08)8:4<164::AID-MFM5>3.0.CO;2-Z. PMID: 10406299.
- Zibell-Frisk D, Jen KL, Rick J. Use of parenteral nutrition to maintain adequate nutritional status in hyperemesis gravidarum. J Perinatol. 1990 Dec;10(4):390-5. PMID: 2126033.
- Peled Y, Melamed N, Hiersch L, Hadar E, Wiznitzer A, Yogev Y. Pregnancy outcome in hyperemesis gravidarum–the role of fetal gender. J Matern Fetal Neonatal Med. 2013 Nov;26(17):1753-7. doi: 10.3109/14767058.2013.798293. Epub 2013 May 24. PMID: 23611752.
- American College of Obstetrics and Gynecology. ACOG (American College of Obstetrics and Gynecology) Practice Bulletin: nausea and vomiting of pregnancy. Obstet Gynecol. 2004 Apr;103(4):803-14. PMID: 15051578.
- Tamay AG, Kuşçu NK. Hyperemesis gravidarum: current aspect. J Obstet Gynaecol. 2011 Nov;31(8):708-12. doi: 10.3109/01443615.2011.611918. PMID: 22085059.
- Madjunkova S, Maltepe C, Koren G. The Leading Concerns of American Women with Nausea and Vomiting of Pregnancy Calling Motherisk NVP Helpline. Obstet Gynecol Int. 2013;2013:752980. doi: 10.1155/2013/752980. Epub 2013 Apr 15. PMID: 23690784; PMCID: PMC3649700.